1 / 3
Gases - Pressure | Volume | Temperature / Gas Laws
Gas Laws
Three variables that affects each other in Gases.
Pressure, Volume and Temperature
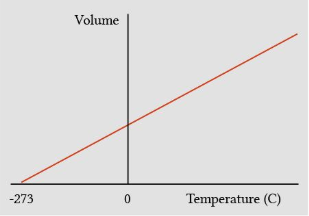
If fixed the Pressure (constant) , Increase in temperature will cause the volume to increase and vice versa
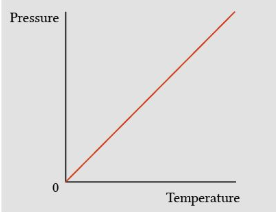
If fixed the Volume (constant, Increase in temperature will cause the pressure to increase and vice versa
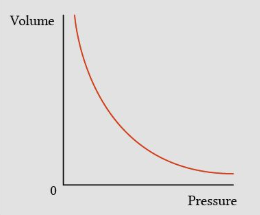
If Temperature is fixed (constant), increase in volume will cause the pressure to decrease and vice versa (BOYLE'S LAW)
Pressure constant
Temperate and volume are directly proportional.
Volume constant
Pressure and Temperature is directly proportional
Temperature constant (Boyle's Law)
Boyle's Law states that when temperature is constant, pressure and volume are inversely proportional.